Dimethyl Ether (DME) Synthesis Process
Recently, Dimethyl Ether (DME) has come to be viewed as a fuel source that will produce clean energy in the future. The features which make DME particularly attractive include the fact that it does not generate any Particulate Matter (PM) as exhaust when used as a diesel fuel substitute, and that it can be easily produced from a number of resources such as natural gas, coal, biomass, plastic wastes or other wastes. The DME synthesis process developed by Mitsubishi Gas Chemical has been applied commercially in small-scale plants. JGC has developed, jointly with Mitsubishi Gas Chemical, an effective process for DME synthesis capable of producing several thousand tons of DME per day. In this technology, the methanol produced from raw materials such as natural gas is treated to synthesize DME through what is called an "indirect process", a means for producing DME by methanol dehydration.
DME Synthesis Process (methanol dehydration)
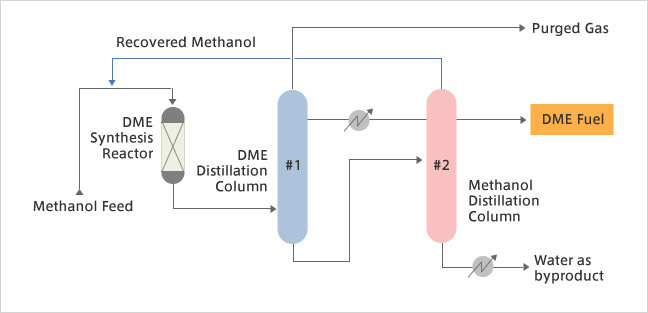
Features
- Applicable to a super-large plant for energy-supply uses
- Employs a high-performance methanol-dehydration catalyst developed by Mitsubishi Gas Chemical
- Accepts crude methanol as feed
- Distillation section is designed to cope with trace amounts of impurities
- A high level of energy efficiency is achieved through a design that maximizes the heat balance in the process
Applications
- DME production from pure and crude methanol feeds
- DME production from methanol produced from biomass, plastic wastes or other wastes via synthesis gas or a mixture of green hydrogen and CO2
Experience
- 80,000 tons-DME per year (Fuel DME Production Company Ltd., Japan)
- 20,000 tons-DME per year (Grillo-Werke AG, Germany)
Energy Transition - Gas/Oil/Chemicals
- The World's Most Advanced Energy-saving Condensate Desulfurization Process(JUSTTM Condensate)
- Integrated Hydrogen Desulfurization System - JUST® Refinery -
- Gas Oil Ultra-Deep Desulfurization Process
- Dimethyl Ether (DME) Synthesis Process
- High-Purity Hydrogen Sulfide Production Process
- DTP® Process
- Advanced Dehydration Process for Organic Compounds (Vapor-Phase PSA System)
- High-Throughput and High-Efficiency Extraction Column (WINTRAY®)